No yoke!
Punnett Squares are a relatively easy way to study certain inherited traits. This activity uses Punnett Squares to determine chicken plumage (feather) colors in certain populations.
For a downloadable Instructor Sheet, Student Instruction Sheet, and Worksheets, click here: PDF of this project
Chicken Plumage Genetics Game
- This material has been oversimplified! For a more thorough discussion on chicken genetics, check out this site.
- It was modified from a Grade 6-12 project but I felt the game would benefit from real-world examples. Blue, for example, is a “real†chicken color. In this project, brown would be more precisely called dun, orange is champagne, and yellow is blonde. The color changes were made as most children aren’t going to be familiar with common poultry plumage colors.
- As for age-appropriateness – my 6-year-old found it far more interesting than did my 13-year-old!
Materials:
1. 2 sets of 6 different colors plastic eggs (per 12 eggs). Suggested colors are given below. 
2. Student Key to put inside eggs: Corresponding colors of candy, buttons, centimeter cubes, or any colored manipulative that will fit, or a slip of sturdy paper with the 4 offspring color marked. If you use candy, you will have to restock each egg, if you use manipulatives or a paper key, you can use it from class to class and year to year.
I used a half-popsicle stick for my key. Egg #6 shows a chick with a blue sire and brown dam. It has a 50% chance of having blue plumage, and a 50% chance of having brown plumage:
You might also notice I numbered everything to reduce future prep time.
3. Worksheet copy for each child. For younger children, color in the phenotype boxes to make this less frustrating.
PDF here: PDF of this project
Instructor Preparation:
1. Introduce age-appropriate concepts of recessive/dominant traits, genotypes/phenotypes, and Punnett squares.
2. The egg shells represent the plumage color contributed by the parents – half the egg represents the Sire (father/rooster), half the egg represents the Dam (mother/hen). Use the genotype/phenotype chart to match plumage color to phenotype.
Simplified Sire/Dam and Offspring Chicken Plumage Genetics:
|
Genotype |
Phenotype |
Use: |
Genotype |
Phenotype |
Use: |
|
BB |
Black Feathers |
1/2 black egg |
RR |
Red Feathers |
1/2 red egg |
|
Bb |
Blue Feathers |
1/2 blue egg |
Rr |
Orange (champagne) Feathers |
1/2 orange egg |
|
bb |
Brown (dun) Feathers |
1/2 brown egg |
rr |
Yellow (blonde) Feathers |
1/2 yellow egg |
3. Inside each egg, place the 4 corresponding colored pieces to show offspring. You can use candy, small manipulatives, or include an answer key (color dots on sturdy paper). Set them up as follows:
|
Sire/Dam |
Geno-type |
Chicks |
Sire/Dam |
Geno-type |
Chicks |
|
black x black |
BB x BB |
4 black |
red x red |
RR x RR |
4 red |
|
black x brown |
BB x bb |
4 blue |
red x yellow |
RR x rr |
4 orange |
|
brown x brown |
bb x bb |
4 brown |
yellow x yellow |
rr x rr |
4 yellow |
|
blue x blue |
Bb x Bb |
1black 2blue 1 brown |
orange x orange |
Rr x Rr |
1 red 2 orange 1 yellow |
|
blue x black |
Bb x BB |
2 blue 2black |
orange x red |
Rr x RR |
2 red 2 orange |
|
blue x brown |
Bb x bb |
2 blue 2 brown |
orange x yellow |
Rr x rr |
2 orange 2 yellow |
4. Demonstrate 1-2 (or more, if necessary) Punnett Squares with the children, then let them do the rest alone or in groups. When they finish each egg, have them open the eggs and check their answers!
This activity can be used to reinforce percentages, as well (ex: 25% of the chicks will have red feathers, 50% will have orange feathers, and 25% will have yellow feathers).
Student Instructions:
Plastic Egg/Chicken Plumage Genetics Game
|
Phenotype |
Genotype |
|
|
Phenotype |
Genotype |
|
Black Feathers |
BB |
Red Feathers |
RR |
||
|
Blue Feathers |
Bb |
Orange Feathers |
Rr |
||
|
Brown (khaki) Feathers |
bb |
Yellow Feathers |
rr |
Directions:
1. Choose one egg, but do not open it yet.
2. One half of the egg represents the plumage (feather) color of the dam (mother/hen).
The other half of the egg represents the plumage (feather) color of the sire (father/rooster).
3. Record the plumage color phenotype and genotype of your egg in the chart.
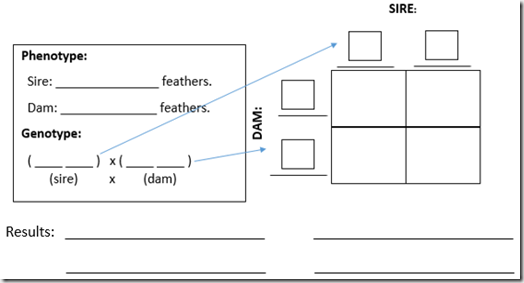
4. Place the genotype of your egg into the Punnett Square.
5. Determine the possible genotypes and phenotypes of the offspring.
6. When you are finished – open your egg! Do your answers match?
a. If yes, put the egg back together, and choose another.
b. If no, check your work, and make corrections
Example of how to fill in Punnett Squares: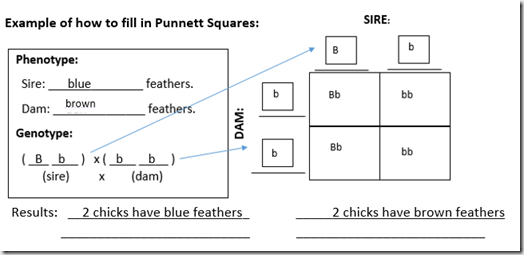
Punnett Squares (blank):